
In recent years, there have been tremendous advances in cutting-edge technologies and scientific methods that promise to complement the use of experimental mammalian models in research, thanks in part to the efforts of NIEHS and other leaders in environmental health sciences. Known as new methodologies (NAMs), these tools range from machine learning and in vitro (cell-based) systems to high-volume chemical screening and sophisticated computational techniques that help scientists integrate and interpret large amounts of toxicology data.
I believe that one NAM that is helping to transform our ability to understand environmental exposures and their potential biological effects is actually a tiny, non-mammalian species – the zebrafish. In early January, I attended an environmental health conference hosted by India’s National Institute of Environmental Engineering, part of the Indian Council of Scientific and Industrial Research. In other presentations, NIEHS grant recipient Dr. Robyn Tanguay of Oregon State University (OSU) discussed her use of zebrafish and why she believes zebrafish represent a paradigm shift in toxicology research.
After the conference, I interviewed Dr. Tanguay (who also directs the NIEHS-supported Superfund Research Center at OSU) to learn more about her scientific portfolio and to better understand what makes zebrafish a potential Game-changing model creatures. We discuss how zebrafish can help researchers study multiple chemicals simultaneously, assess the impact of gene-environment interactions, prioritize substances studied in traditional animal models, and uncover key biological mechanisms that may be altered by exposure. I also asked Dr. Tanguay what inspired her to become a scientist.
Zebrafish Research Benefits
Rick Wochik: Why are zebrafish an ideal model for studying potential health effects of environmental exposures?

Robin Tangay: The genetic similarities between zebrafish and humans are remarkable because the genes required to make humans are very similar to those required to make fish. By leveraging the similarities and other properties of zebrafish, researchers can integrate this model organism in important ways, including more quickly identifying important exposures that interact with our genetic and biological makeup.
Zebrafish are native to India and they have different side stripes, similar to zebras. Adult worms are about an inch in length, but most of our work is actually done during the embryonic development stage, when we’re talking lengths in millimeters. In the early stages of life, they are transparent, so you can actually watch the development of the brain, heart, eyes, ears, etc. using a simple, inexpensive microscope. For example, this makes it easy to see how exposure affects the development of specific organs, among other benefits.
Another major benefit of studying zebrafish is their rate of development. We can start with one cell one morning and by noon the next day, the heart is already beating and many other organ systems are formed in just two days. This makes our research faster and more efficient compared to traditional rodent systems. The rapid development of zebrafish – combined with the different imaging capabilities and genetic analysis tools at our disposal – is allowing us to make exciting discoveries at scale and at low cost.
Importantly, nearly every gene encoded in an organism is required to perform one or more tasks during early development. Sometimes a gene is only temporarily needed to complete a developmental task, and then later in life it may play an important role in the adult liver, for example. But we know that nearly all genes are expressed and active during embryonic development. Therefore, this is an ideal time to see whether chemicals disrupt gene function and cause observable physical changes that are associated with disease outcomes.
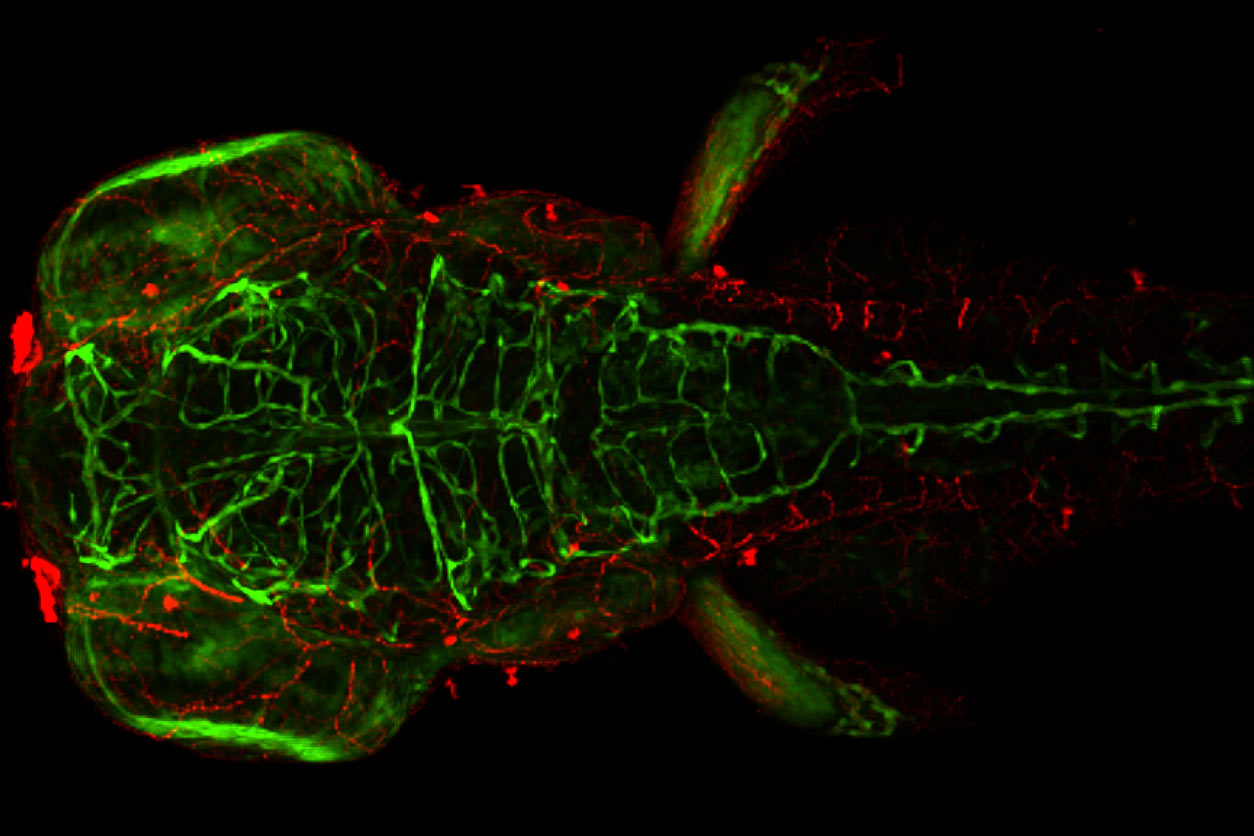
A breakthrough in neurobehavioral assessment
RW: For years, your lab has used zebrafish to study the potential health effects of chemicals such as polycyclic aromatic hydrocarbons, flame retardants, pesticides, and complex mixtures. Can you expand on this work for our readers?
RT: certainly. We first sought to observe the normal course of zebrafish development to understand their baseline physical characteristics and behavioral attributes. This may include heart rate, eye diameter, body curvature, and more. Next, we started using different categories of individual chemicals for exposure, such as dioxins, metals, and other chemicals you mentioned. The goal is to identify all abnormal phenotypes resulting from exposure and then study them more deeply by analyzing the genetic and biological mechanisms involved.
One thing we noticed right away is that many different chemicals can cause common physical and behavioral effects, such as cardiac edema (swelling caused by fluid accumulation). By itself, this information does not give us insight into the underlying mechanisms involved, but it does show the value of our model in screening physical and behavioral changes induced by various exposures, which will help to prioritize Our follow-up assessment.

Furthermore, we found that we can use zebrafish to study neurobehavioral effects on a large scale, which was previously not possible and quite challenging in cell-based models. This means we can quickly assess changes in nervous system development, changes in motor activity, etc. We then attempted to perform a comprehensive assessment, measuring all phenotypes at multiple concentrations over several days. Many of my group’s early innovations in this area came from NIEHS funding, and the challenge was to do something that might represent a paradigm shift in the field. I think our work has had that effect and I’m grateful for the support.
trace cause and effect
RW: How does your team move from identifying a phenotype to better understanding the genetic and biological mechanisms that may be causing it? How does this knowledge translate to the human environment?
RT: In the field of toxicological risk assessment, there is increasing interest in identifying gene expression changes that predict disease outcome and then determining the exposure levels associated with that outcome. This is the same scientific knowledge we gain from zebrafish research. We used transcriptomics (the large-scale assessment of changes in gene expression in an organism) to determine which genetic changes may result from lower exposure levels that are more like what humans experience in daily life.

When we identify these gene expression changes, we can try to see which changes are adaptive and which are maladaptive. As humans, we are exposed to different substances all the time, but our bodies adapt and respond by changing metabolism or eliminating chemicals and then restoring homeostasis. When you reach these tipping points—either destroying too many genes or modulating the activity of genetic processes and altering homeostasis—disease begins to set in.
By looking at exposures that both affect gene expression and produce a phenotype, we hope to identify a causal relationship—to show that the exposure not only causes the phenotype, but that the phenotype is caused by a specific genetic mechanism. The gene-editing techniques we have at our disposal allow us to powerfully test which aspects of the genetic landscape may play a role in the phenotypes we observe. Additionally, due to the small size and transparency of zebrafish, we can observe in real time how cells interact with each other after exposure and changes in gene expression, which further aids our analysis.
When it comes to using knowledge from zebrafish research in decision-making settings, I know regulatory agencies are now receiving zebrafish data as part of their information packages. I work closely with the EPA (U.S. Environmental Protection Agency) and FDA (U.S. Food and Drug Administration) to help them become more comfortable with this new data flow. I view the exposure-specific data in the zebrafish model as a case study through which we can identify correlations with traditional research data and, more importantly, try to understand why disagreements may exist, which may help strengthen the Use of models. I believe that going forward, policymakers will use zebrafish data to help make more informed choices that better impact public health.
Clarify PFAS
RW: Speaking of human-related exposures, I understand that your lab has begun studying a substance that has been making headlines in recent years—per- and polyfluoroalkyl substances, or PFAS. Can you share your initial work in this area?
RT: The diversity of these structures is remarkable. We’ve screened hundreds of them in zebrafish systems, and I can say they definitely behave differently. Some can bioaccumulate but others cannot, some are metabolized and others are active at low concentrations. So we’re trying to use our model to better understand the properties of these chemicals that make them more or less dangerous.
We want to identify PFAS that are bioaccumulative and have specific biological targets, which are the most feared types of molecules. Substances that are bioaccumulative but have no specific target may become a problem with long-term exposure, but I’m not as worried about those as I am about substances that are capable of achieving a biological target and modulating it. We hope our zebrafish system can help determine which PFAS are safe to use and which should be phased out.
(To learn more about Tanguay’s zebrafish research and latest publications, visit her lab website.)

